GARWOOD MEDICAL DEVICES
Preventing orthopedic implant infection with disruptive UB technology
Garwood Medical Devices' approach to infection control is through a minimally invasive medical device that uses a low-voltage electrical current with a goal to kill antibiotic-resistant bacteria on metal implants. Patented by the University at Buffalo and Syracuse University and exclusively licensed by Garwood Medical, the technology behind the device served as the catalyst for a multifaceted, collaborative partnership between the life sciences company and the university.

How Garwood Medical collaborates with UB
CHALLENGE & OPPORTUNITY
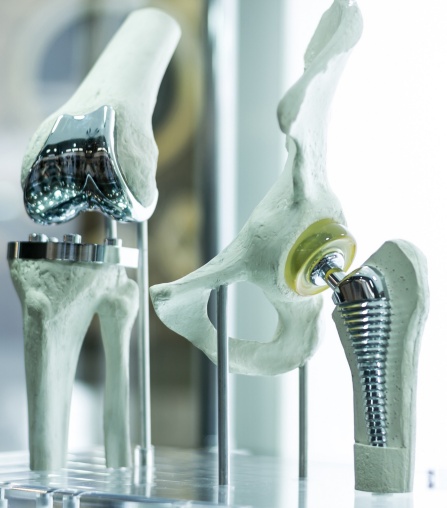
Devastating impacts of implant infection
Each year in the United States, hundreds of thousands of people suffer from orthopedic implant infections that are extremely difficult to cure. In some cases, antibiotic-resistant bacteria form a slime or “biofilm” on an implant. The film acts as a barrier that shields the harmful bacteria from antibiotics or the patient’s immune system.
Current treatment options are painful and invasive, typically involving surgery to remove the infected implant. This costs patients and the healthcare system tens of billions of dollars per year and often still results in joint fusions, amputations and deaths. Treatment innovations could cure these serious infections and preserve implants, saving limbs and lives.
SOLUTION & OUTCOME
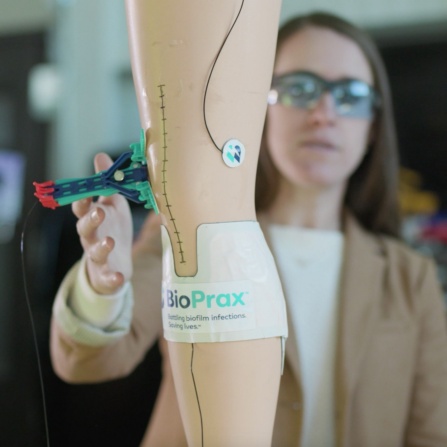
Innovation could lead to better infection control
Technology licensed from UB and Syracuse University is the driving force behind Garwood Medical’s BioPrax™— a minimally invasive medical device that aims to prevent and treat orthopedic implant infections without the need for implant removal. When doctors detect a biofilm infection, BioPrax™ uses a needle to deliver a low-voltage electrical stimulation to the metal implant, which has been shown in extensive pre-clinical testing to break up biofilms, kill bacteria and will target infection prevention in the future.
BioPrax™ is expected to reduce patient morbidity and mortality, prevent unnecessary surgeries, retain implants, help maintain biomechanical stability and mobility and save billions of dollars per year worldwide.
UB SUPPORT
Accelerating commercialization
Beyond licensing multiple technologies from UB, Garwood Medical leverages UB’s supercomputing facility and expertise to perform high-fidelity simulations to analyze heat transfer, electrochemistry and blood flow data. These types of collaborative R&D projects inform current and future design improvements and optimization.
With multiple research projects jointly funded by UB, the company has moved BioPrax™ from proof of concept in a petri dish to large animal surgical safety and efficacy testing in a Good Laboratory Practice (GLP) preclinical research center at UB.
Garwood Medical’s partnership with UB helped them gain FDA Breakthrough Device status and win millions of dollars in funding. In turn, this allowed them to recruit expert biomedical engineering graduates from UB to support R&D efforts, expansion and revenue growth.














