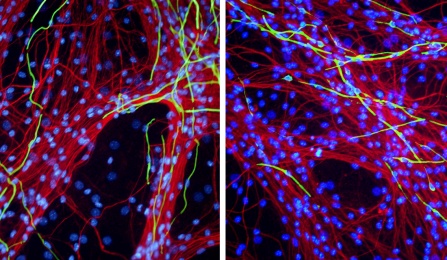
Cultured Dorsal Root Ganglia neurons (in red) myelinated by Schwann cells (nuclei in blue, myelin in green).
Former Feltri Laboratory
The Feltri lab seeked to understand how cell-cell and cell-extracellular matrix interactions contribute to cell shape and polarity within a developing tissue. They applied these biological questions to the development of the nervous system and to the process of myelination, with the goal to translate basic findings into a cure for demyelinating diseases.
The Feltri laboratory closed on 1/31/2025. In an effort to preserve Dr. Feltri's scientific legacy, we have preserved this page which includes the research she was working on at the end of her career, plus a legacy page that summarizes her entire career.
On this page:
Feltri Research Overview
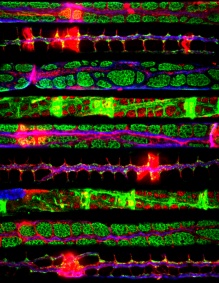
Sciatic nerve fibers that were teased and immunostained for various components of Cajal Bands and of the cytoskeleton.
The Feltri lab research focused on adhesion between myelinating cells, axons and the extracellular matrix and the signals that promote myelination. One of their major efforts was the study of laminin receptors on Schwann cells, the myelinating peripheral glia. By generating and comparing animal models of demyelinating neuropathies to patient's biopsies, we and others have determined that laminins are required for 'radial sorting' of axons in early development. Radial axonal sorting is a pre-requisite for myelination and is arrested in human laminins and dystroglycan-glycosyltransferases deficiencies. Using conditional mutagenesis we have determined that integrins, dystroglycan and RhoGTPAses are required for radial sorting because they induce cytoskeletal rearrangements that allow the generation of glial extensions that contact and wrap axons.
We have adapted innovative sub-fractionation and proteomic techniques to profile the extensions contacting axons and the RhoGTPAse interactome in Schwann cells. By these techniques we have identified novel molecules important for myelination and for the support of axons by glial cells. We have recently discovered that mechanical forces generated by the extracellular matrix and other cells are also essential for correct myelination, and we are actively pursuing the molecular mechanisms by which mechanical signals are transduced in myelin-forming glia.
Laminin receptors are also important for myelin and nodes of Ranvier to achieve the correct length, thickness, architecture and stability. Patients lacking laminins have abnormally thick and instable myelin, short internodes and immature nodes of Ranvier. We first determined that laminin 211, dystroglycan and certain integrins are required to form myelin of normal cytoarchitecture, and now we are seeking to understand why perturbing laminin function leads to myelin instability and demyelination.
Using genetic, cell biology and biochemistry we tested the hypothesis that laminin receptors influence growth factors and signaling molecules to prevent demyelination. These studies also lead us to discover that some of the signaling molecules under scrutiny are more important in the central nervous system, where they inhibit oligodendrocyte myelination. Thus, they represent potential molecular targets to promote remyelination in demyelinating disease such as Multiple Sclerosis of Leukodystrophies.
While at the Institute for Myelin and Glia Exploration, the Feltri laboratory researchers applied their experience on conditional mutagenesis to ask if there is cell autonomy in the pathogenesis of Krabbe disease.
Dr. M. Laura Feltri
DIRECTOR, 2011 - 2023
SUNY DISTINGUISHED Professor of Biochemistry & Neurology
Dr. Feltri was a Professor of Biochemistry and Neurology at the Institute for Myelin and Glia Exploration in the State University of New York at Buffalo and Director of the Institute. Before 2011, she was the Head of the Unit of NeuroGlia in the San Raffaele Scientific Institute of Milano, and adjunct Associate Professor in the Department of Neurology at the University of Pennsylvania.
Past Members
- Kansho Abiko, Undergraduate
- Dominique Ameroso, Master's Student & Research Technician
- Kathleen Catignas, PhD Student
- Narayan Dhimal, PhD Student
- Kevin Espino, Undergraduate
- Iman Faraj Musbah Fergani, Master's Student, Neuroscience
- Scott Ferguson, Undergraduate
- Monica Ghidinelli, PhD Student
- Arsalan Haghdel, Undergraduate
- Yoochan Hwang, Undergraduate Student & Research Technician
- Leandro Marziali, Postdoctoral Associat; Research Scientist
- Kenneth Minorczyk, Undergraduate
- Seth Moore, PhD Student
- Clementine Namba, CLIMB UP student & NIH Diversity Supplement awardee
- Gustavo Della Flora Nunes, Undergraduate
- Marilena Palmisano, PhD Student
- Marta Pellegatta, PhD Student
- Yannick Poitelon, Postdoctoral Research Scientist
- Thomas Rush, Undergraduate
- Oliver Simpson, Undergraduate
- Sri Laxmi Veerapaneni, Master's Student, Neuroscience
- Jordan VerPlank, Postdoctoral Research Scientist
- Michael Weaver, MD-PhD Student
- Nadav Weinstock, MD-PhD Student
- Emma R. Wilson, Postdoctoral Research Scientist
- Irene Yu, Medical Student
- Jiajun Zhang, Research Technician I
